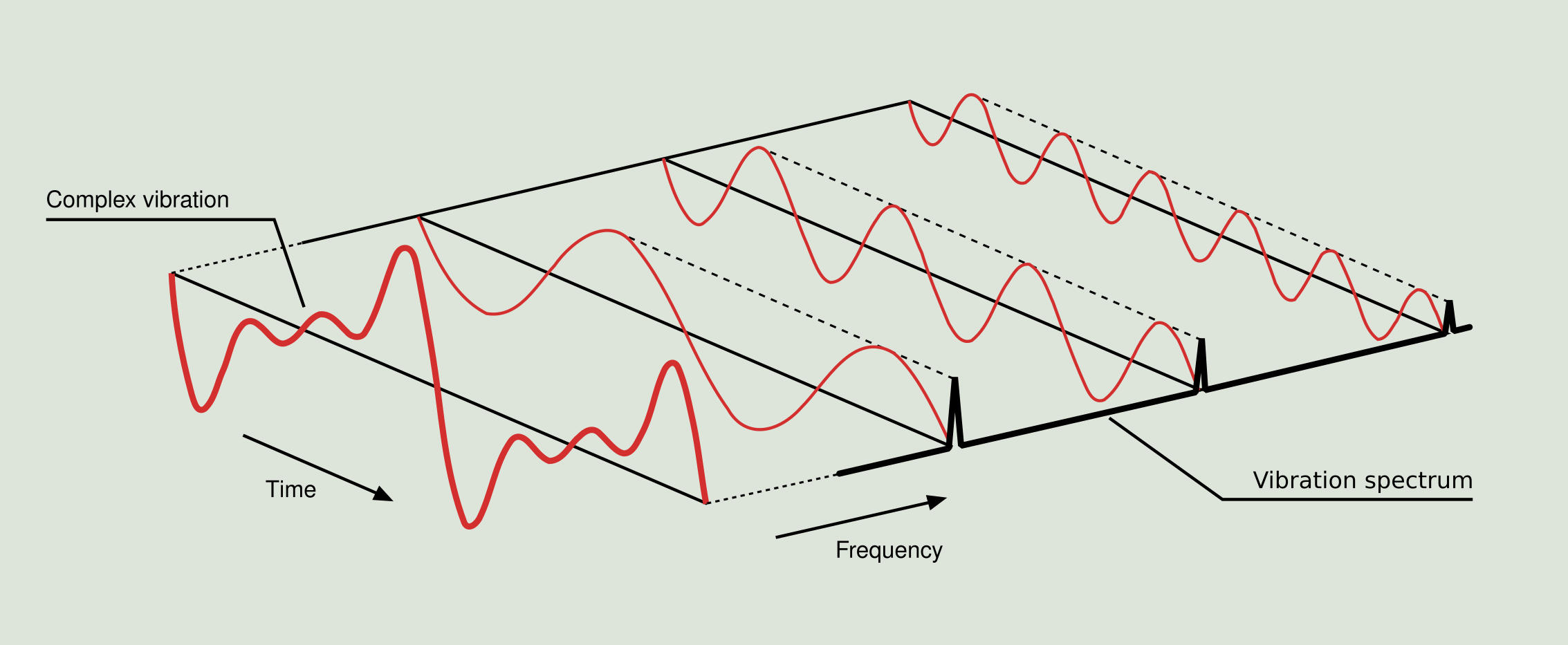
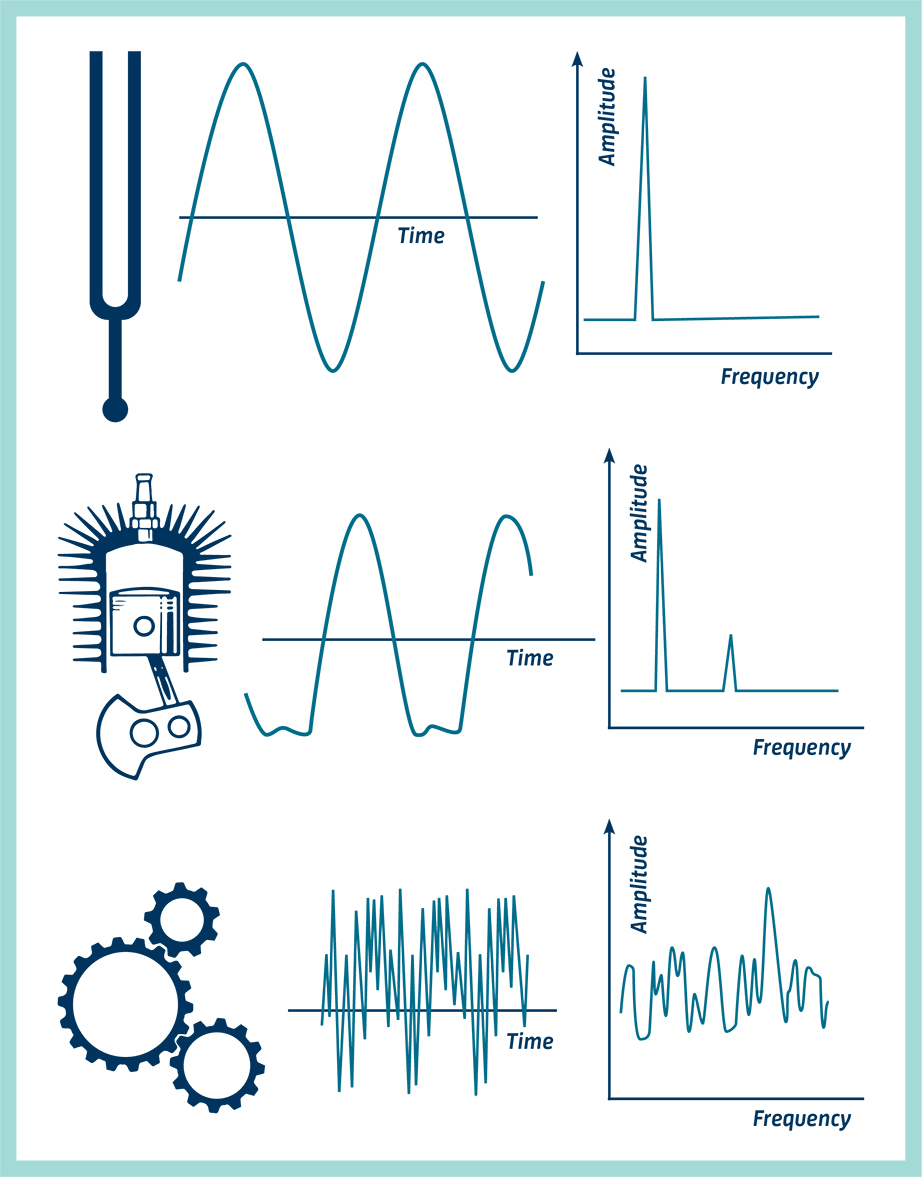
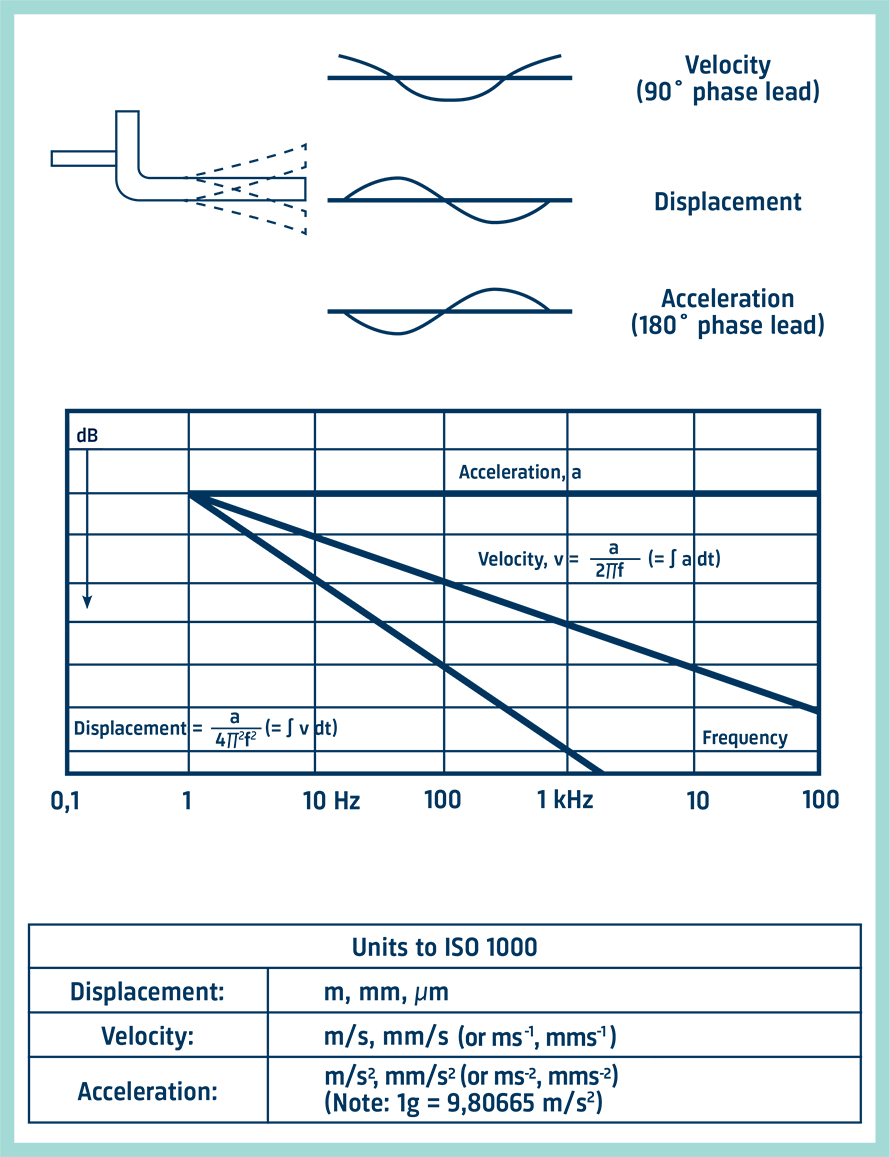
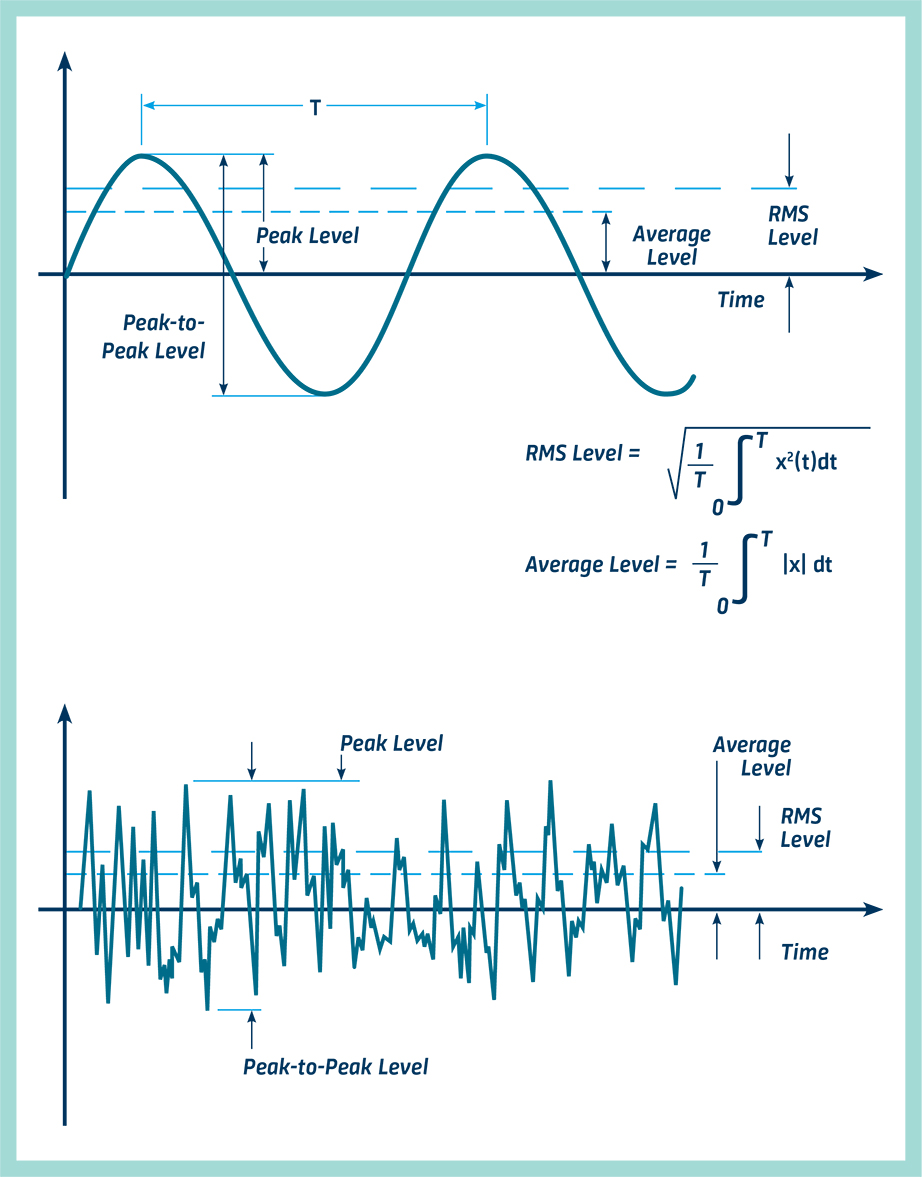
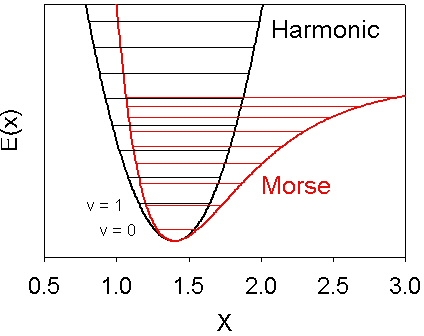
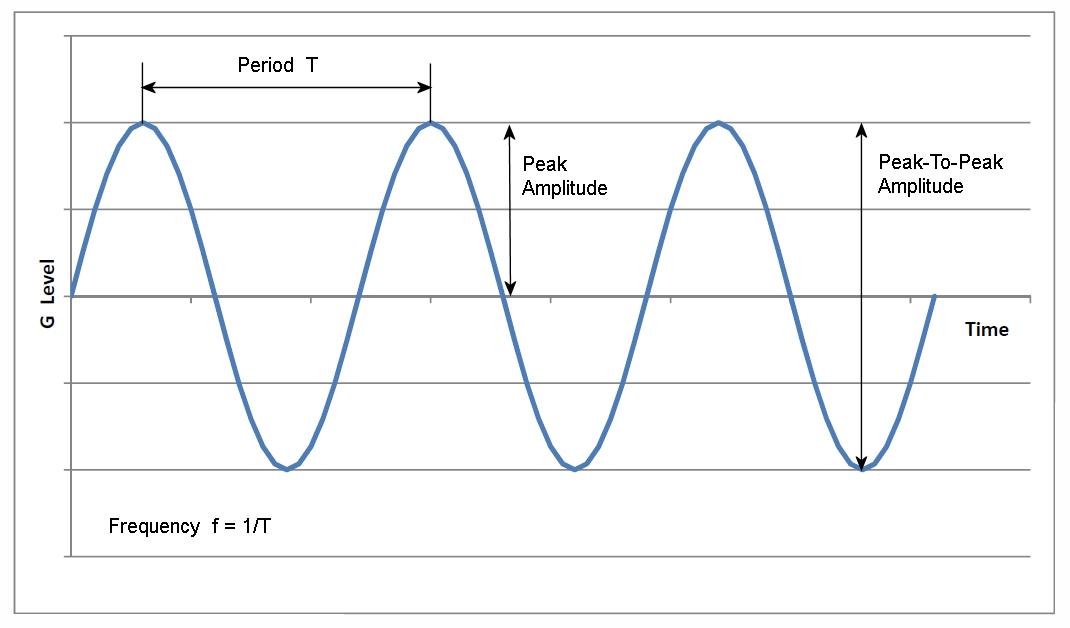
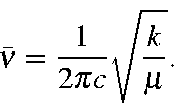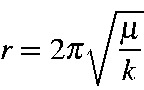Vibrational energy. The vibrational energy (in arb. units) is plotted... | Download Scientific Diagram
How do I calculate the force constant, zero-point energy, and the energy level spacings for ""^(12) "C"""^(16)"O" if tildeomega_e = "2170 cm"^-1? | Socratic

SOLVED: The vibrational energy levels for diatomic molecule with natural frequency are given by: E, (n + H)hv Calculate, in units of eV and kJ mol" the energy level spacing for: chlorine

Calculation of Vibrational Frequency - Introduction to Organic Chemistry - Lecture Notes | Study notes Organic Chemistry | Docsity

SOLVED: The following equation; derived from Hooke' law for vibrating spring; expresscs vibrational frequency a5 wavenumber (cm"'): v(cm-!) =4.12 k= force constant of bond (dynes cm-!) # (reduced mass) (atomic mass units)
![The frequency of vibration of the string is given by v = p2l [ Fm ]^1/2 Here, p is the number of segments in the string and l is the length. The The frequency of vibration of the string is given by v = p2l [ Fm ]^1/2 Here, p is the number of segments in the string and l is the length. The](https://i.ytimg.com/vi/4gO3FfK5AE0/maxresdefault.jpg)
The frequency of vibration of the string is given by v = p2l [ Fm ]^1/2 Here, p is the number of segments in the string and l is the length. The

OneClass: Part a? Consider the H2 molecule: The force constant of H2 is k = 510 N/m. Calculate the fu...