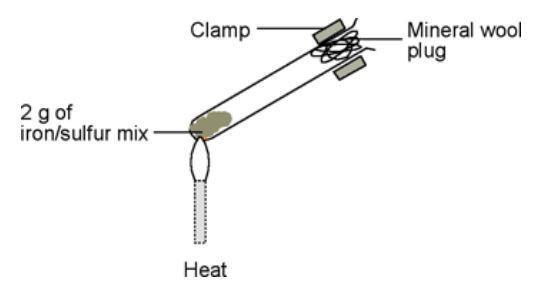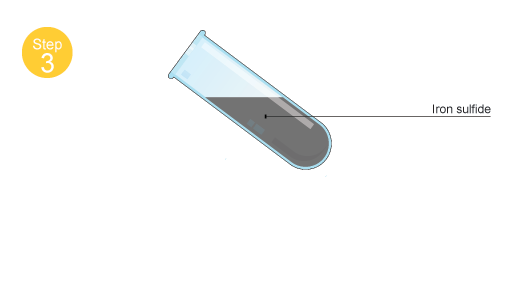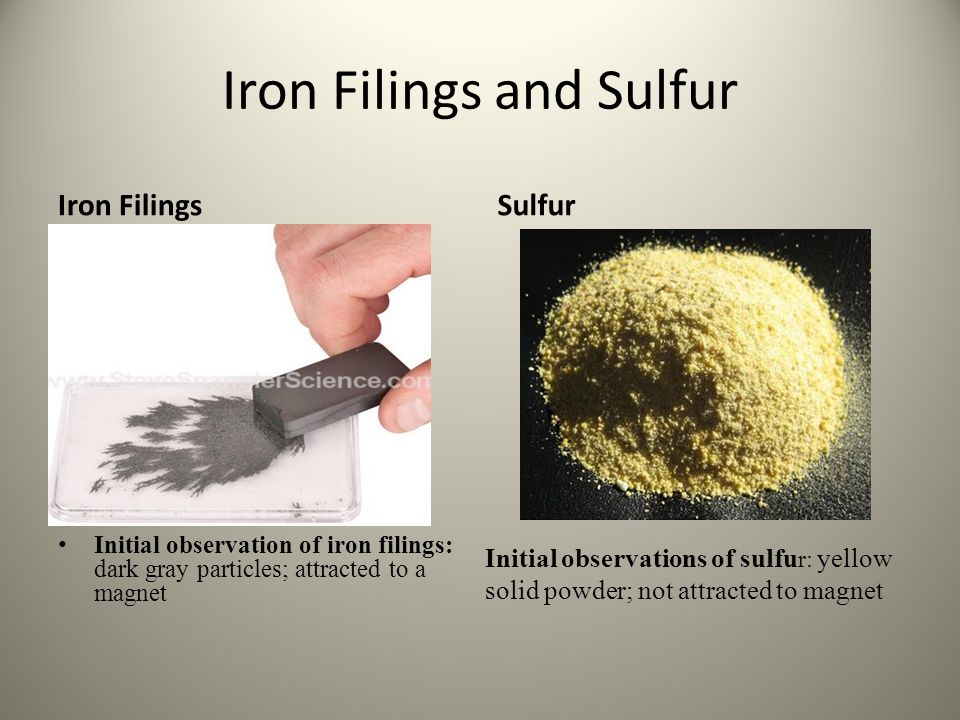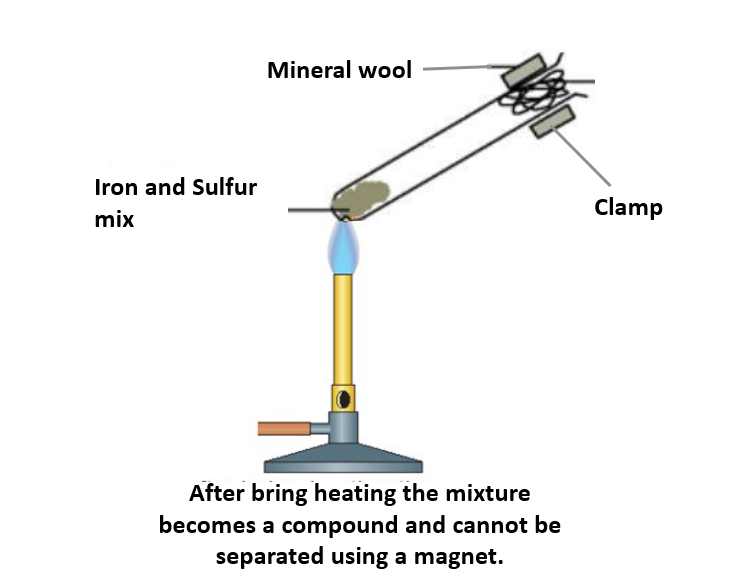
What is formed if a mixture of powdered iron and sulphur is heated in a test tube?A.An elementB.A mixtureC.A compoundD.None of the above
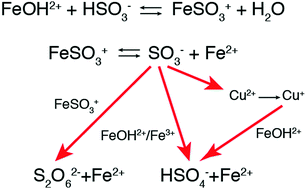
The oxidation of sulfur(iv) by reaction with iron(iii): a critical review and data analysis - Physical Chemistry Chemical Physics (RSC Publishing)

Iron reacting with sulphur in a test tube. This is an addition reaction that produces iron sulphide. The equation for this reaction is: Fe + S ---> Fe Stock Photo - Alamy






:max_bytes(150000):strip_icc()/iron-pyrite-cubic-crystals-in-matrix--collected-at-mina-victoria--navajun-la-rioja--spain-578457937-5ad8a68a18ba010037eeae61.jpg)