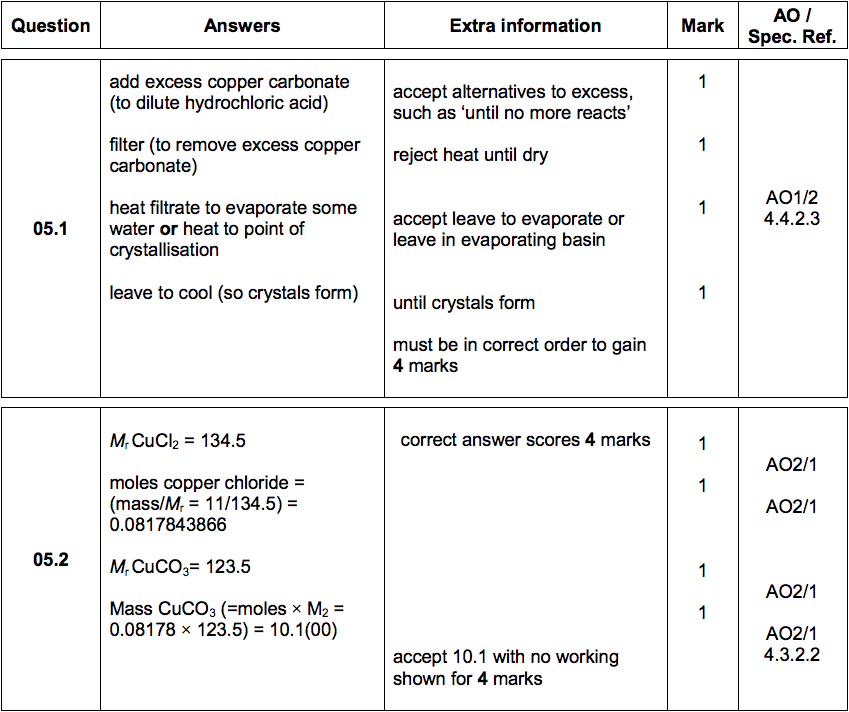
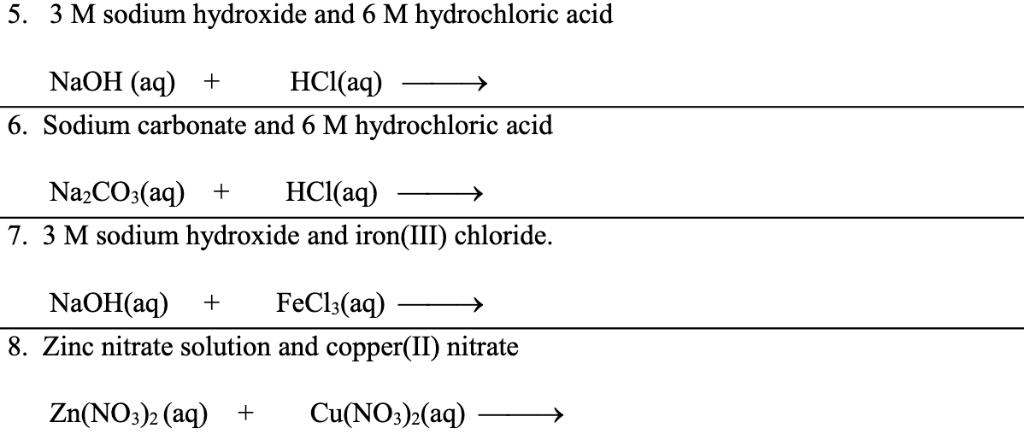
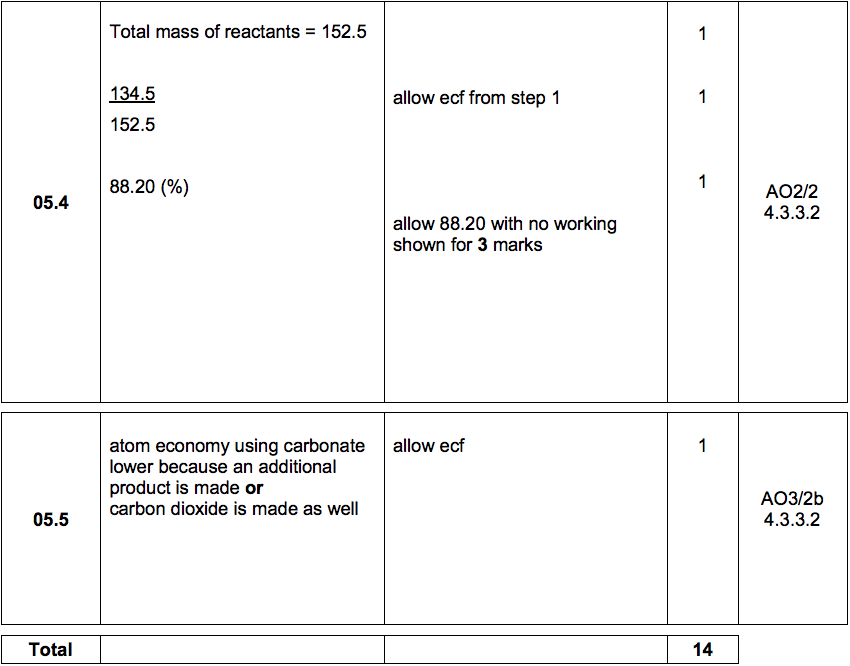
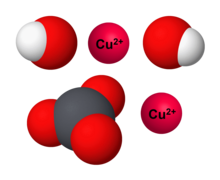
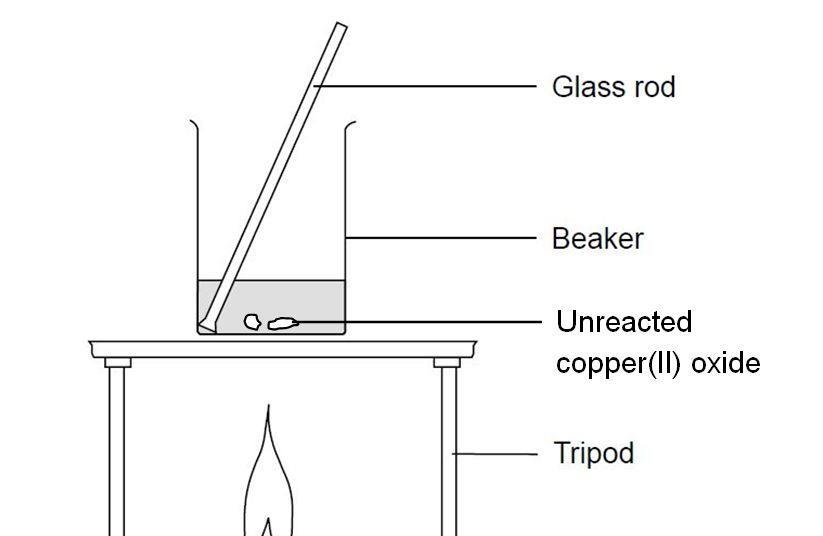
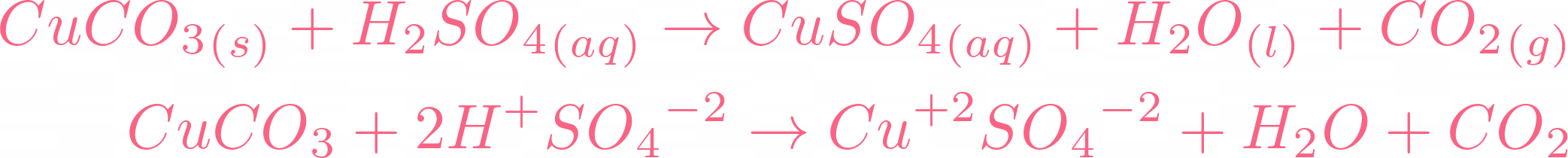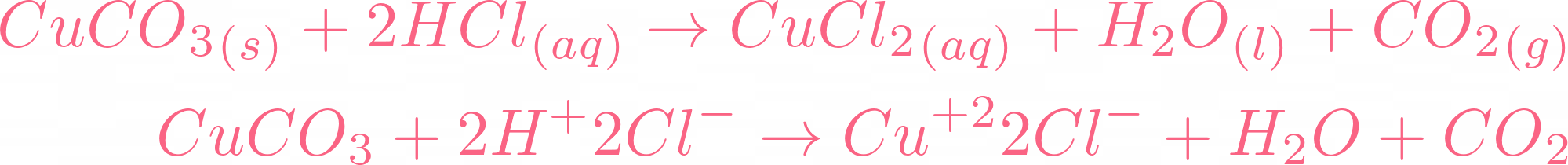7. 0 g of copper (ll) carbonate is added to 50cm3 of 1.0mol dm-3 hydrochloric acid. What is the mass of copper (ll) carbonate left at the end of the reaction? - Quora

Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution
Dilute hydrochloric acid is added to copper carbonate. - Sarthaks eConnect | Largest Online Education Community

Copper Carbonate,Senior Chemistry - Extended Experimental In-Industry News-Nickel Acetate,Cobalt Sulfate-Fairsky Industrial Co., Limited