Reaction of chalk and acid. This is an example of an acid-carbonate reaction. Chalk contains calcium carbonate, and the acid here is hydrochloric acid Stock Photo - Alamy

Chapter 15 slides - S1 - Lecture notes 15 - Chap. 15: Chemical Kinetics Powdered chalk (mostly - Studocu

Bildagentur | mauritius images | Reaction rates. Reaction rate increases with concentration of reactants. This effect is demonstrated here using the reaction of chalk (calcium carbonate, CaCO3) with hydrochloric acid (HCl). Carbon

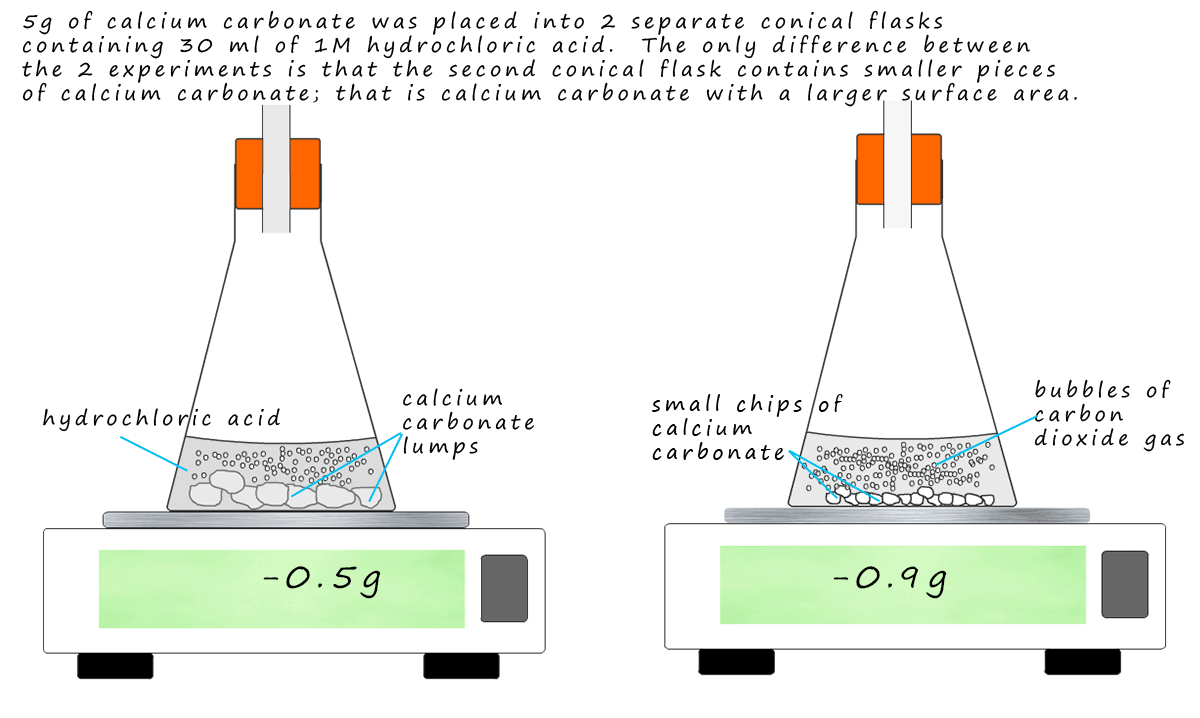
Write a balanced chemical equation the reaction of calcium carbonate and dil. hydrochloric acid.{ CaCO }_{ 3 }+2HClrightarrow { CaCl }_{ 2 }+{ CO }_{ 2 }+{ H }_{ 2 }O{ CaCO }_{