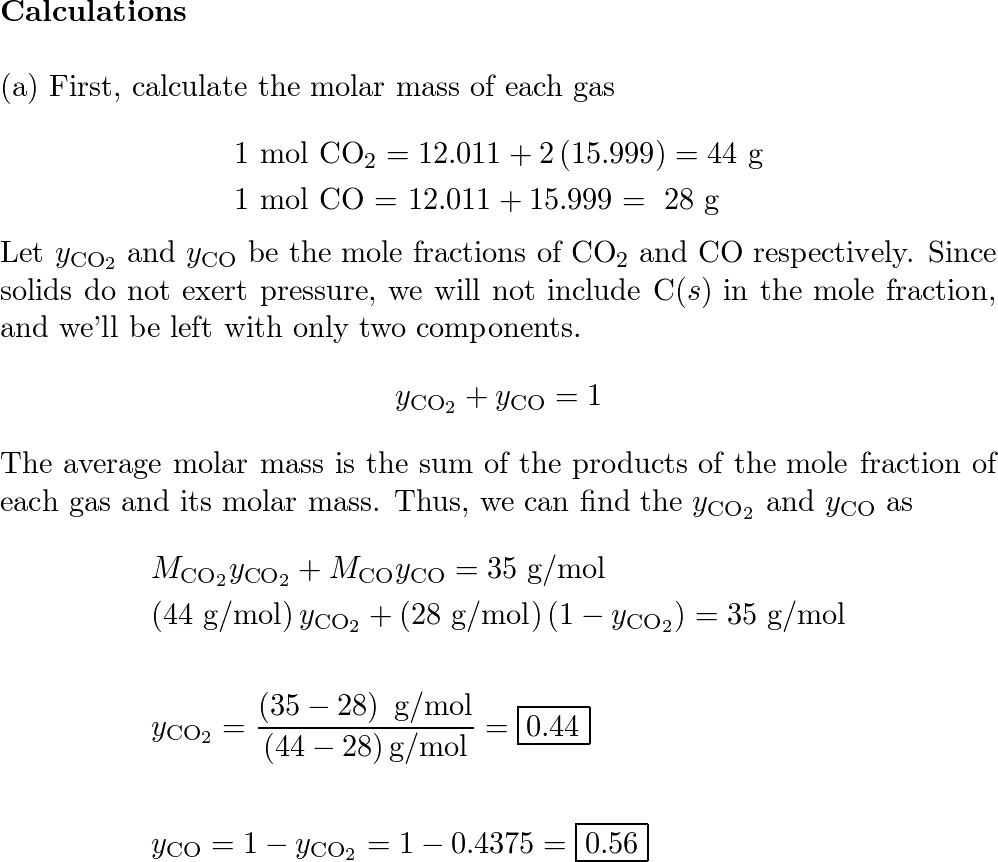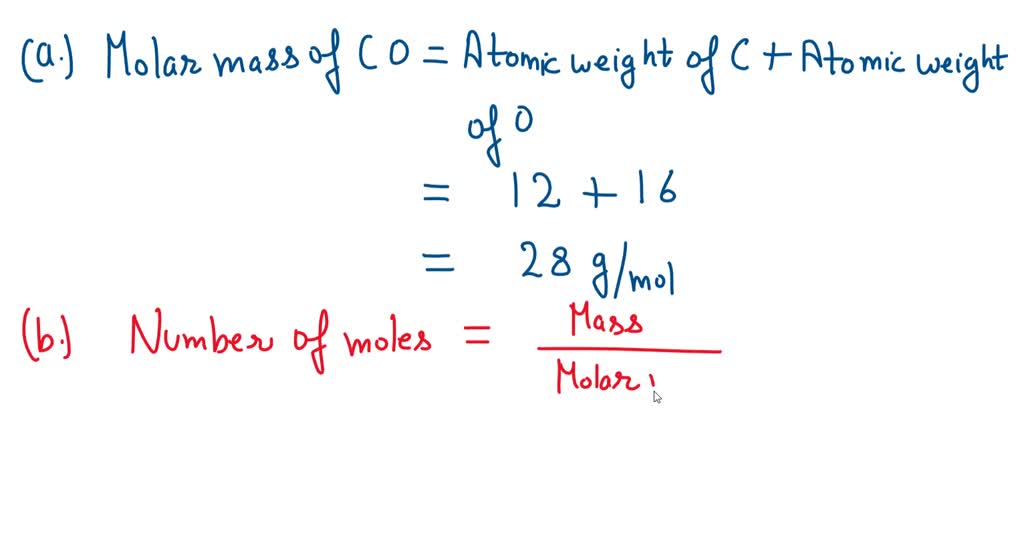
SOLVED: What is the molar mass of carbon monoxide (CO)? Atomic weight of carbon is 12 and oxygen is 16. Answer: g/mol How many moles of CO gas if its mass is
XHCO3 and YCO3 Are two pure substancesof equal molar masses decomposing as shown 2XHCO3===>H2O +CO2 +X2CO3 YCO3===>YO + CO2 16.8g of XHCO3 gave 6.2 g of mixture of water and carbon dioxide