
4: SWNTs showing hexagonal structure of the carbon atoms and covalent... | Download Scientific Diagram
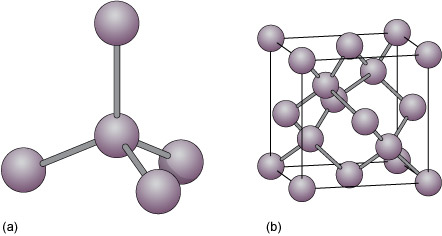
Minerals and the crystalline state: 4.3 Covalent structures and bonding | OpenLearn - Open University
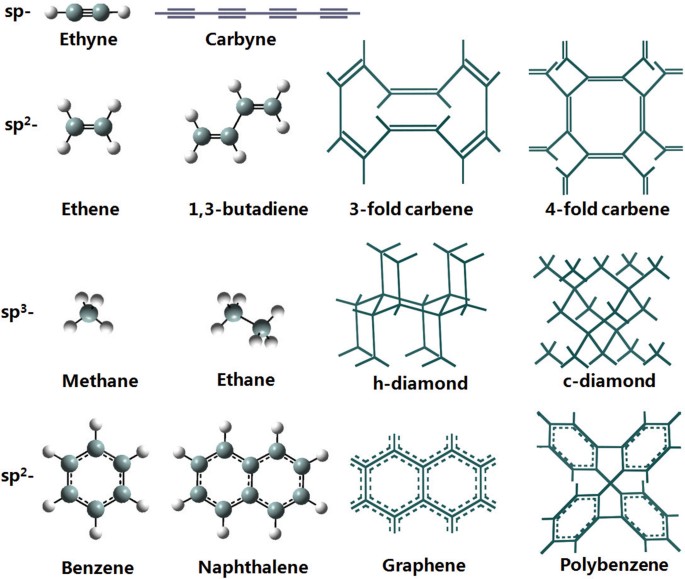
New Carbon Allotropes with Helical Chains of Complementary Chirality Connected by Ethene-type π-Conjugation | Scientific Reports
Giant covalent structures - Structures - (CCEA) - GCSE Combined Science Revision - CCEA Double Award - BBC Bitesize



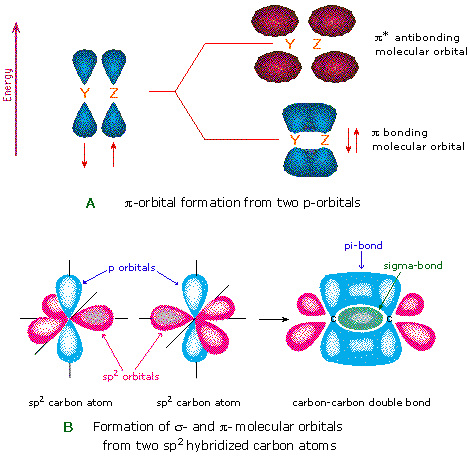
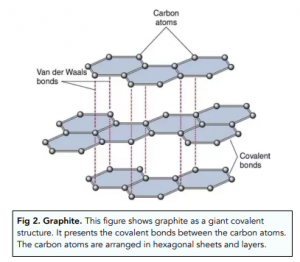
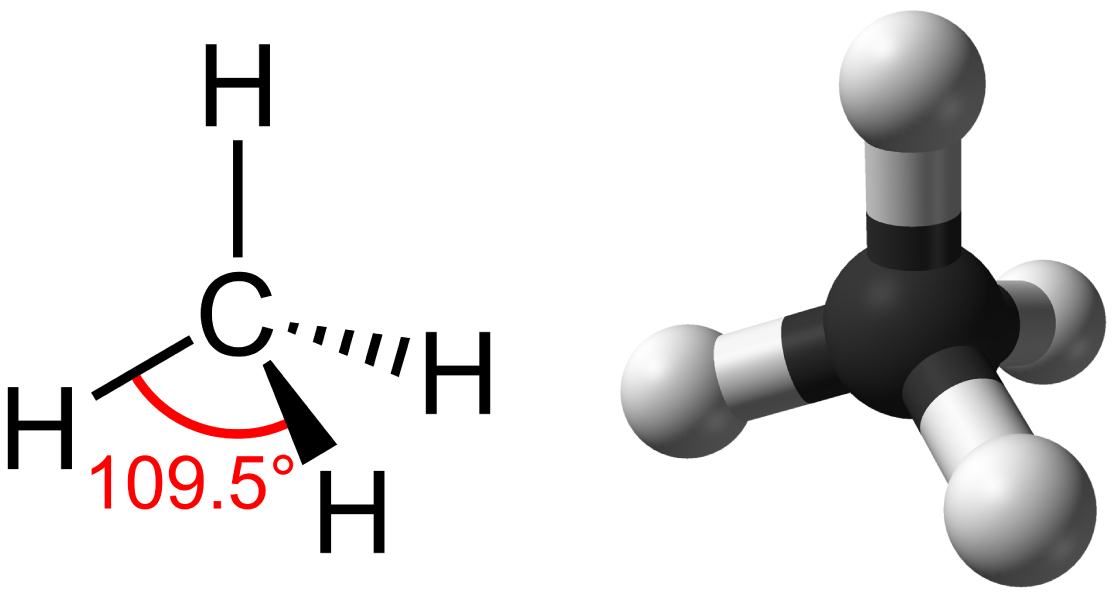



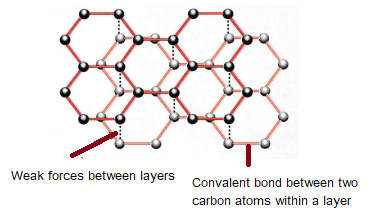




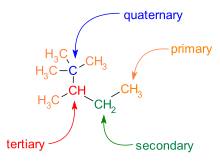

%202.3.2.1%20-%20The%20structure%20of%20graphite-min,h_400,q_80,w_640.jpg)