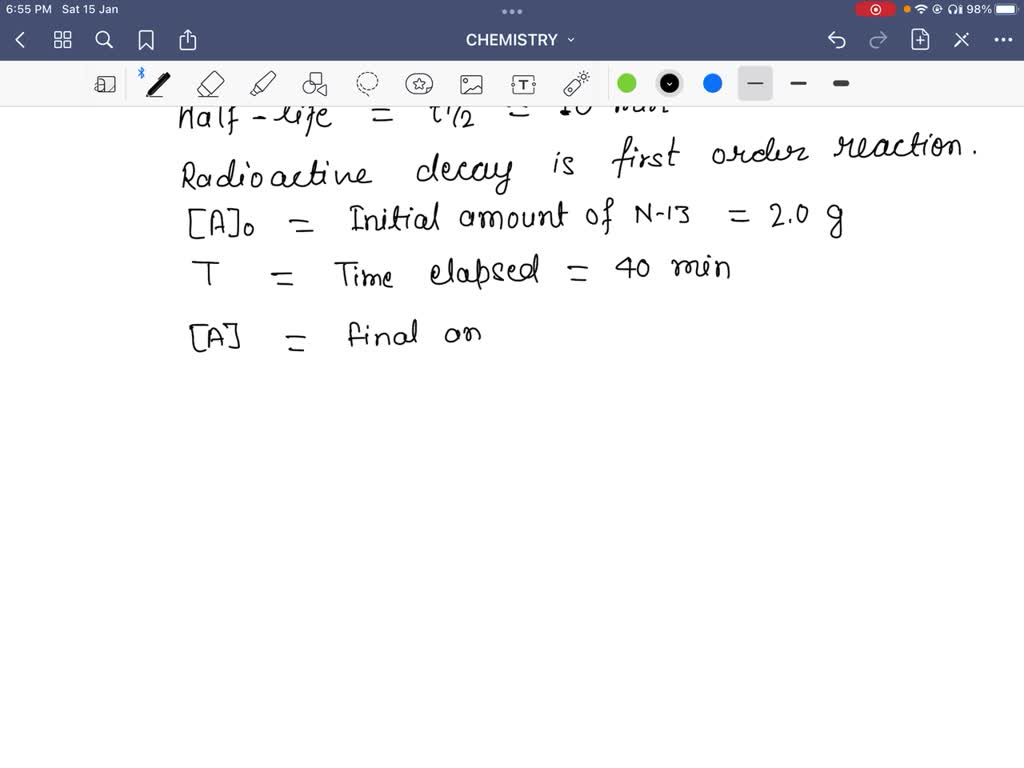
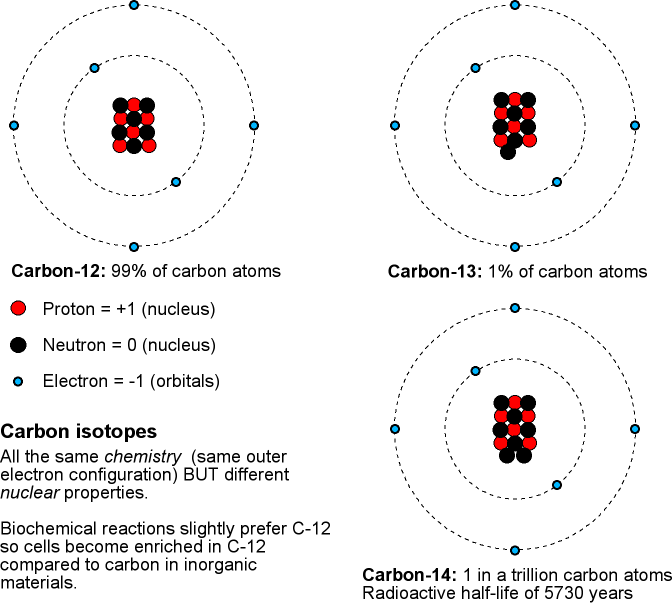
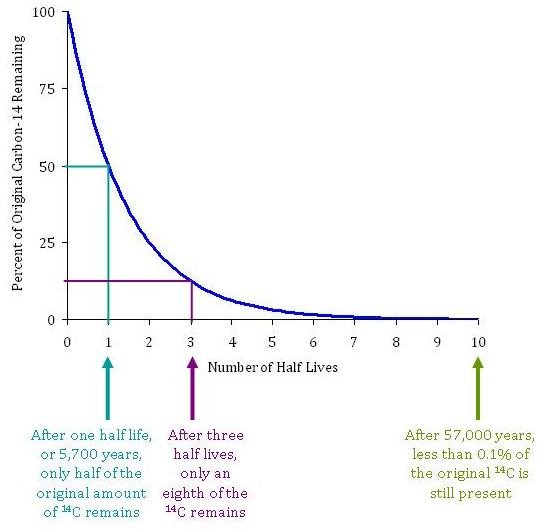
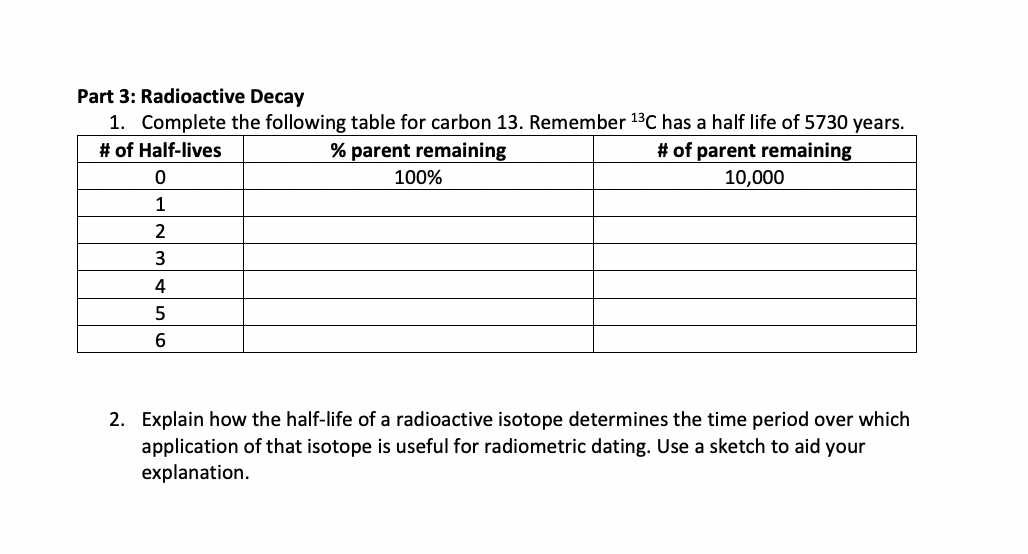
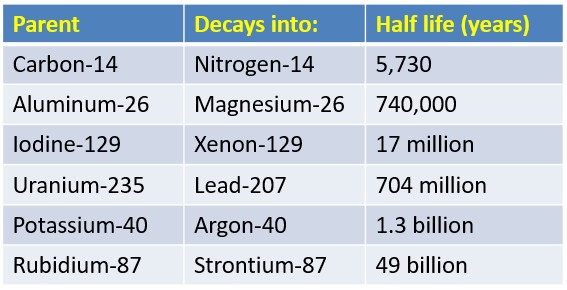
Is the half-life of Carbon-14 always EXACTLY 5,730 years? Is it possible for the half-life of a carbon-14 sample to be 5,728 years? Or 5,732 years? - Quora

ABSOLUTE AGE Absolute Dating Radiometric Dating Half Life Isotope Radioactive decay Carbon ppt download
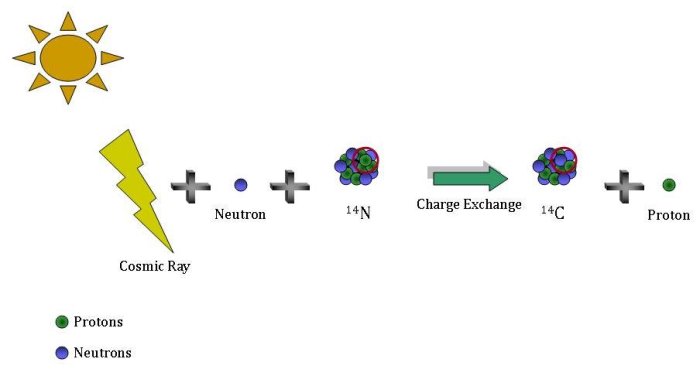
How do we know that the half life of carbon 14 is 5730 years? And how does finding out the ration between c12 and c14 pertain to radiocarbon dating? - Quora
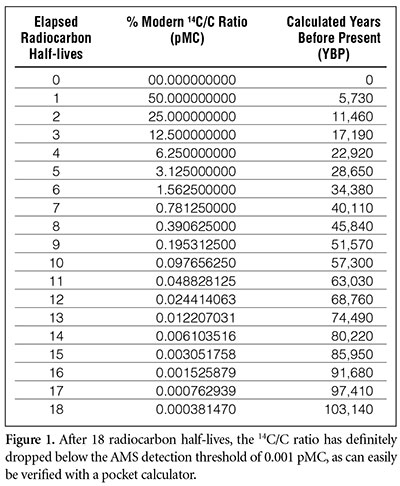
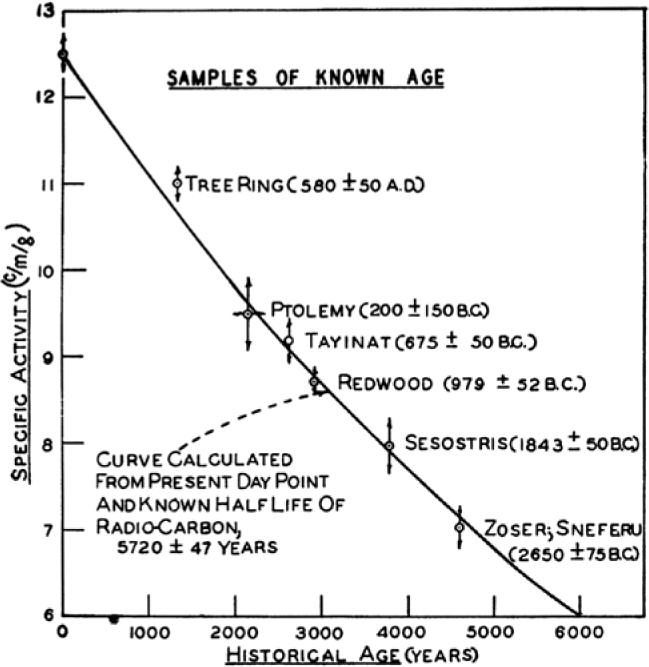
Rethinking Carbon-14 Dating: What Does It Really Tell Us about the Age of the Earth? | The Institute for Creation Research

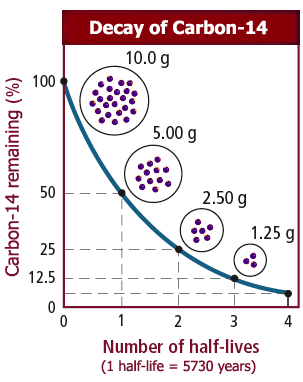
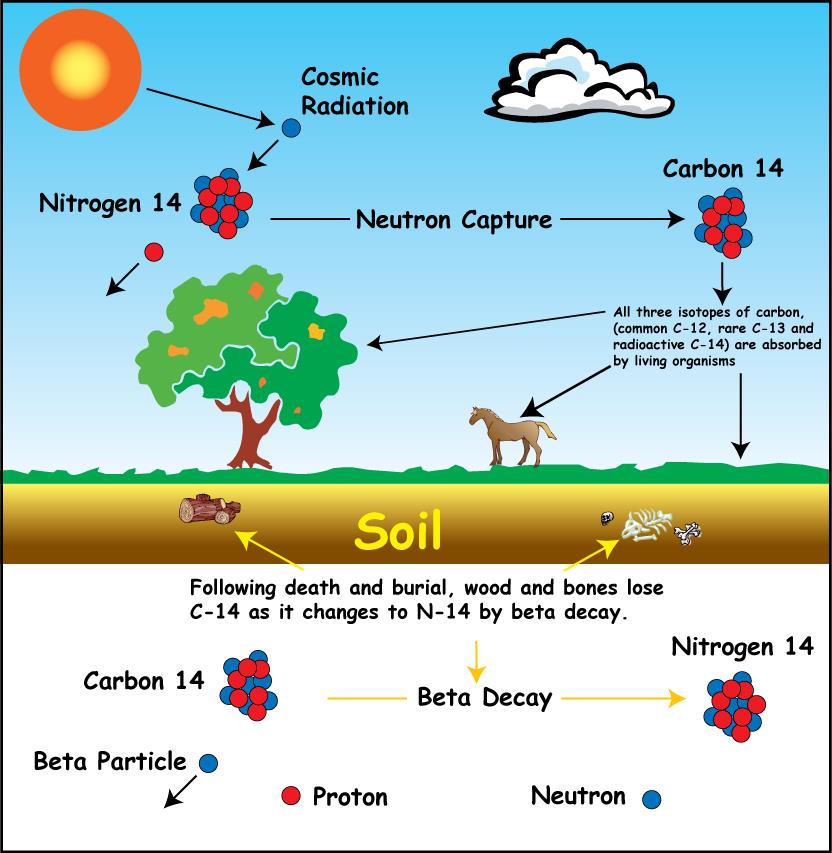
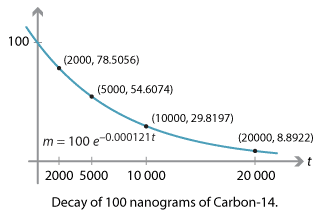
6. How long is material radioactive? carbon-14 archaeological dating, half- life uses, decay curve, geological dating of igneous rocks, radiocarbon-14 dating, long term storage problems of high level nuclear waste KS4 science igcse/gcse

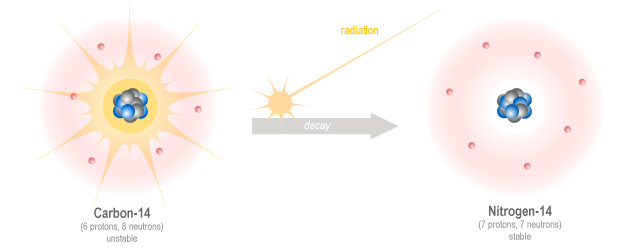
When Cows and Kids Collide: Mythical Creation Science: Doesn't Carbon-14 Dating Disprove the Bible? Part 1
What would happen if all the carbon atoms in the human body were replaced with a heavier isotope? - Quora