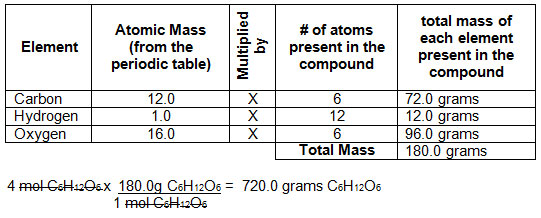
Calculate the amount of carbon dioxide that could be produced when 1 mole of carbon is burnt in air. 1 mole - Brainly.in
30. Calculate the amount of CO2 that will produce. 1)For 1 mole of C is burnt in 16 g of O2 and 2)For 2 mole of C is burnt in 16 g of O2
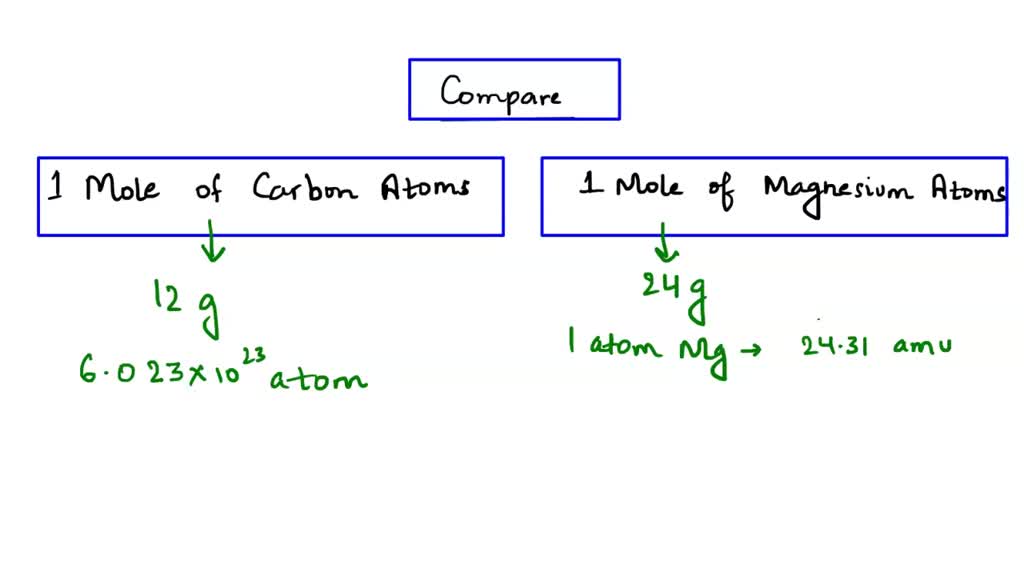
SOLVED: In comparing 1 mole of carbon atoms to 1 mole of magnesium atoms, which statement is TRUE? The mass of 1 mole of carbon is greater than the mass of 1